ESR1 mutation detection in liquid biopsy samples
GALEAS™ uPCR: ESR1 (UKCA)
A qPCR assay with the sensitivity of a digital PCR assay designed for detecting eleven key mutations in the estrogen receptor gene, ESR1.
Accuracy and sensitivity of digital PCR in a qPCR assay
GALEAS uPCR: ESR1 detects eleven of the most prevalent mutations in the estrogen receptor gene, ESR1, known to be associated with endocrine therapy resistance. The assay uses ultrasensitive PCR (uPCR), a Nonacus proprietary technology, to deliver improved sensitivity and specificity over standard qPCR that ensures no wild-type bleed through and data comparable to digital PCR.
Simple to use, and requiring no specialist equipment, it offers laboratories a fast and cost-effective method for the accurate and sensitive detection of ESR1 mutations.
Mutations in the ESR1 gene are known drivers of resistance to standard endocrine therapy and are present in up to 40% of ER+, HER2- metastatic breast cancers1. Liquid biopsy studies have led to a greater understanding of the role these mutations play in resistance and clinical guidelines increasingly support the use of ESR1 testing in metastatic breast cancer2.
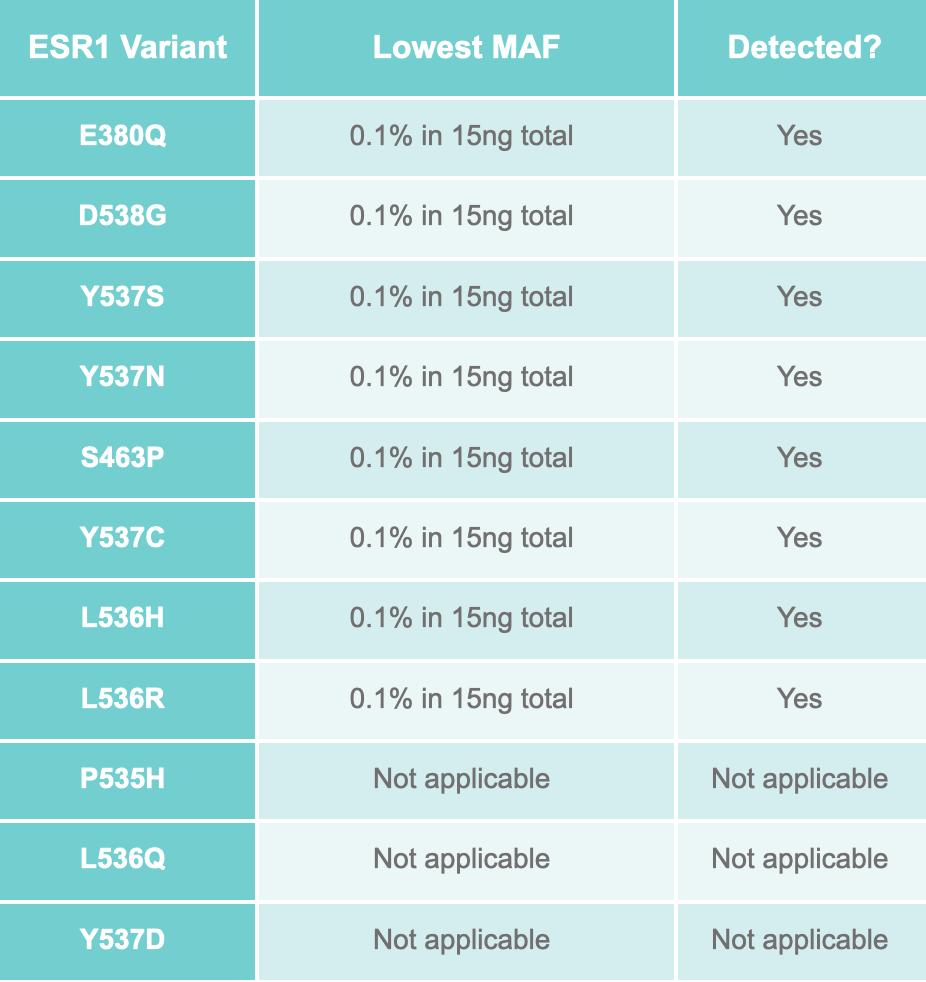
ESR1 variants covered by GALEAS uPCR: ESR1:
High prevalence: E380Q, D538G, Y537S, Y537N
Low prevalence: S463P, Y537C, L536H, L536R, P535H, L536Q, Y537D
Key features
- Detects eleven of the most prevalent mutations in the estrogen receptor gene, ESR1, known to be associated with endocrine therapy resistance.
- Developed for use with cell-free DNA.
- Delivers improved sensitivity and specificity over standard qPCR.
- Simple to use, requires no specialist equipment.
Performance
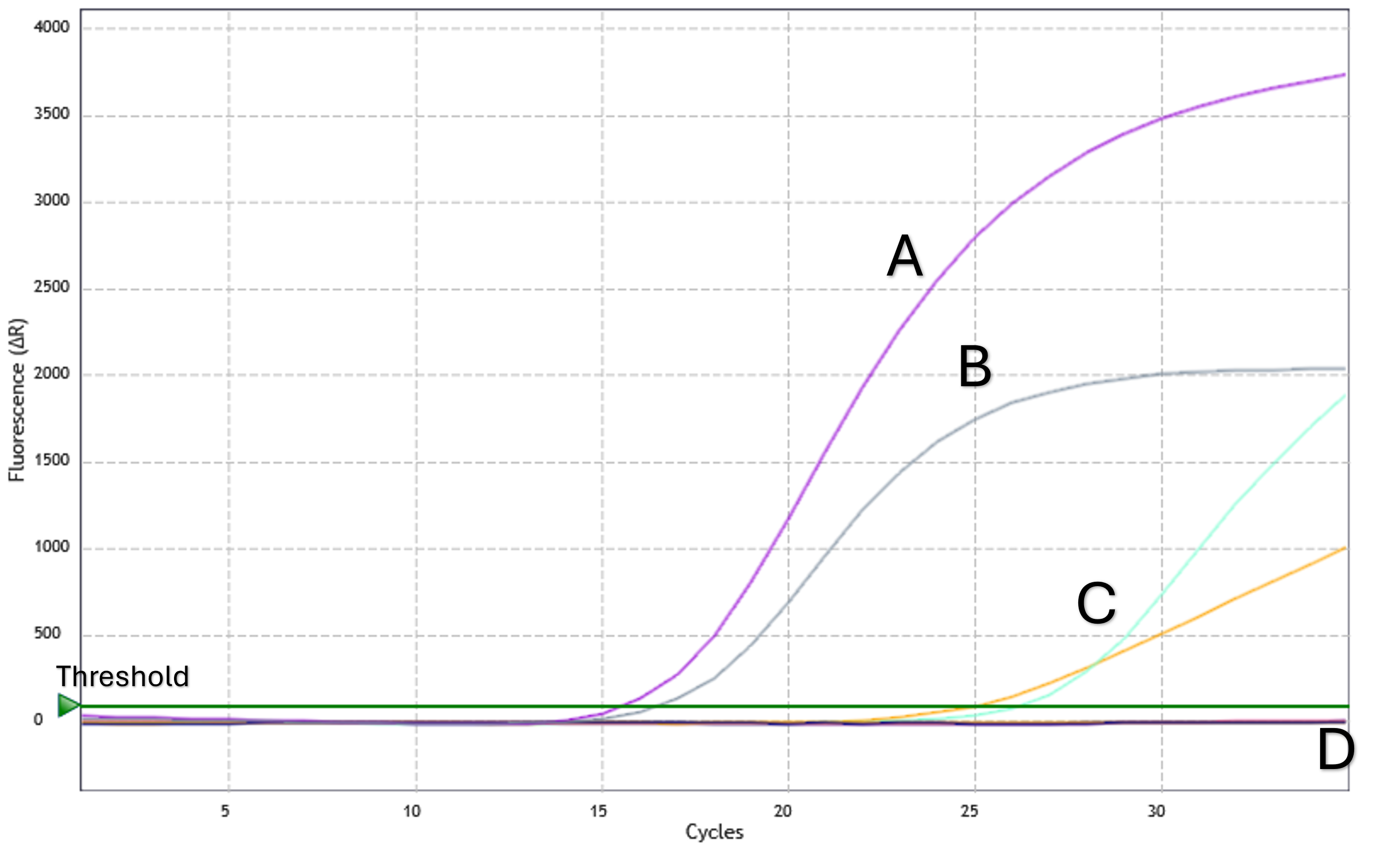
Assay sensitivity and specificity were tested using commercially available reference standards and synthetic controls containing multiple, known ESR1 variants.
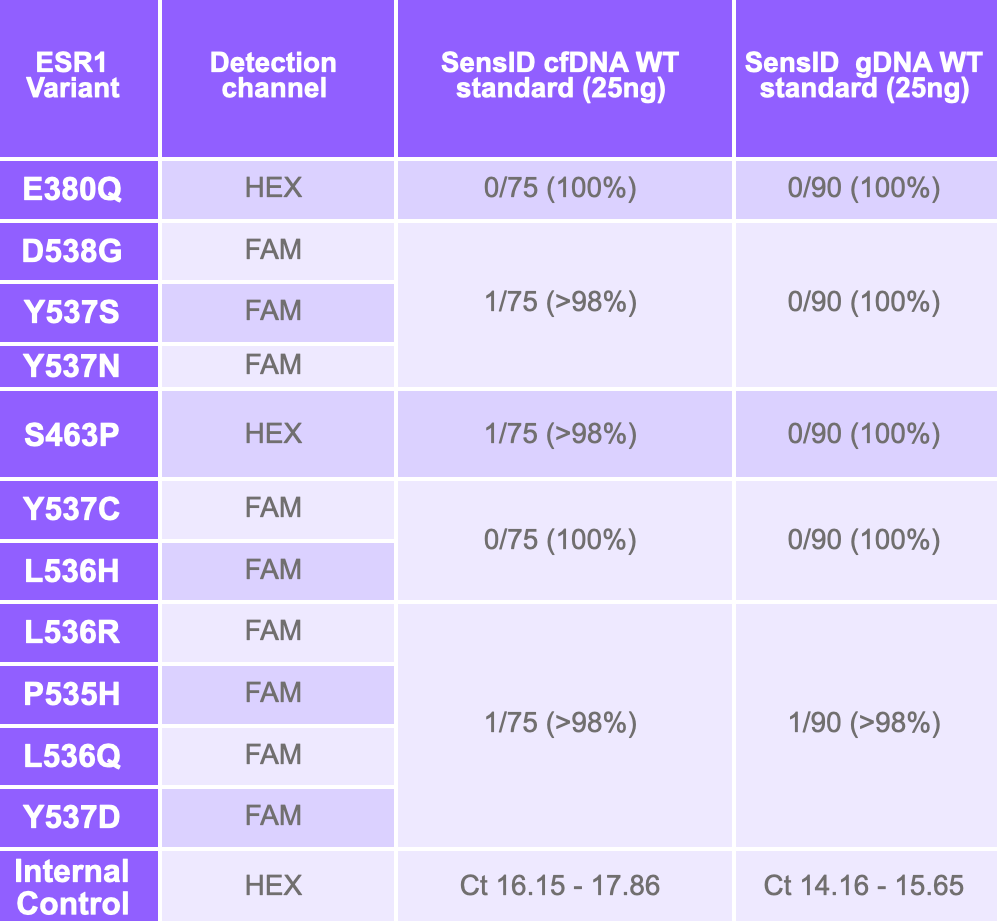
The assay was validated using the ESR1 reference set from SensID, GmbH at 1%, 0.3% and 0.1% MAF in a background of SensID wild-type cfDNA. 0.1% MAF was detected, and 100% specificity was achieved.
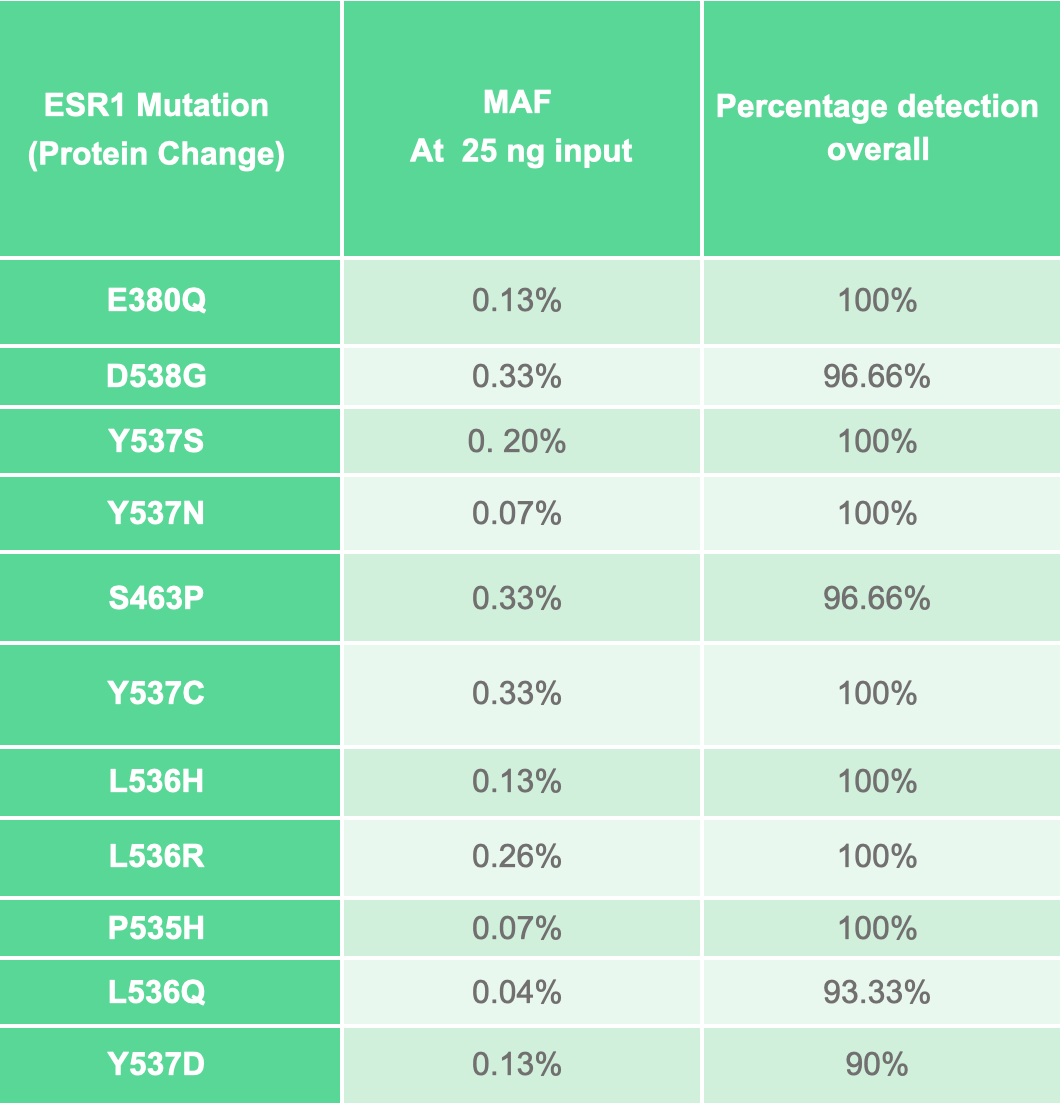
Analytical sensitivity and specificity were confirmed using synthetic controls. ESR1 mutations were detected with a specificity of ≥98% and a sensitivity of ≥90% at mutant allele frequencies (MAF) between 0.04% and 0.33%.
Streamlined workflows; quick and easy protocols
Nonacus uPCR
Sample to data in under 3 hours:

Sample Preparation

PCR Amplification and fluorescence read out

Analysis
Why choose GALEAS uPCR: ESR1?
Accurate and sensitive
Detect mutations in ESR1 with a specificity of ≥98% and a sensitivity of ≥90% at MAFs between 0.04% and 0.33%.
Key variants covered
Detect 11 actionable variants in the ESR1 gene from cell-free DNA in plasma
Results in under 3 hours
A fast, simple qPCR protocol that requires no specialist equipment and can be deployed in almost any molecular laboratory.
GALEAS uPCR: ESR1 is UKCA marked in accordance with the UK Medical Devices Regulations 2002 (as amended). Nonacus Ltd. is the legal manufacturer and is responsible for the development and ascertainment of the performance characteristics of GALEAS uPCR: ESR1.